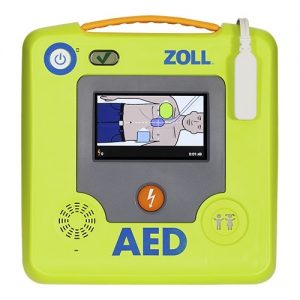
 ZOLL AED 3
ZOLL AED 3
ZOLL AED 3 BLS
- This AED package includes:
- ZOLL AED 3 BLS semi-automatic AED
- CPR Uni Padz
- Sealed Lithium Manganese Dioxide Battery
- Carry Bag
ZOLL AED 3
ZOLL AED 3 BLS
The Zoll AED 3 BLS is a semi-automatic AED for First Responders who are Basic Life Support trained. Because the sensors of the electrodes measure the speed and depth of chest compressions and adjust the rescuer accordingly, the quality of CPR increases. It has one ECG waveform which can be seen on the color display screen.
The Zoll AED 3 adjusts (if necessary) at the speed and depth of the chest compressions, the chances of survival of the victim of a cardiac arrest increase enormously. The quality of CPR largely determines the chance of survival.
The defibrillator has an Ingress Protection value of 55. This value indicates how well the AED is resistant to dust and water. The AED 3 can be placed in dusty rooms due to the high IP value.
With the push of a button you can change resuscitation protocol, so that the Zoll AED 3 semi-automatic can also be used for a child’s resuscitation. Fortunately, children’s resuscitations are rare. Less than 5% of all CPR consists of child CPR.
Because the defibrillator is a semi-automatic device, a button must be pressed if the AED orders a shock. The AED indicates this by means of a voting command. After the rescuer has pressed the shock button, the AED delivers the shock at the best moment.
The lifespan of the electrodes and battery of this AED is 5 years, the AED 3 has low maintenance costs.
The reliability of a defibrillator often depends on its self-testing. As you may imagine, an AED is (fortunately) rarely used.
ZOLL AED 3 Features
Special Features ZOLL AED 3
- With Real CPR Help and CPR Dashboard
- High Resolution ECG display
- “Real Time” CPR feedback on CPR Rate and Depth
- CPR Cycle Timer
- Integrated Voice, text, and visual prompts
- Wifi and USB Connectivity – automatically transmit device status to program management via wifi and push case data (Program free in the first year)
- 8 years warranty on the AED unit.
Please call 0917-1696611
Or contact us via email form below
Other AED Brands

Mindray
Mindray develops and manufactures medical devices and accessories for human and veterinary use. The company exists of three business lines: Patient Monitoring & Life Support, In-Vitro Diagnostic Products and Medical Imaging Systems.

ZOLL Medical
ZOLL Medical Corp, a company of Asahi Kasei is a well-known player in the resuscitation industry. For almost 40 years they develop medical devices and software solutions.

Philips
Koninklijke Philips N.V. is a Dutch multinational conglomerate corporation, originated in Eindhoven, Netherlands. Philips is organized into two main divisions: Philips Consumer Health and Well-being and and Philips Professional Healthcare.

Physio Control
Physio-Control (Stryker), one of the leaders in the development, manufacturing, sale and service of external defibrillators, patient transport and data solutions. Well-known for their Heartsine AEDs and Lifepak defibrillators. Acquired by Stryker Corporation In 2016.

Cardiac Science
Cardiac Science, 100+ years of existence and a well-known leader for AEDs and diagnostic cardiac monitoring devices, providing clinicians with effective tools to identify and treat heart disease, assess results and help improve public health.

Radian
Radian Q Bio is established in 2005 in Korea. Their business activities are research, development, manufacturing of sensors, medical devices and measuring equipment. Since 2014 Radian included the Heart Guardian AED series to their product portfolio.
